Subutex—the brand name for buprenorphine—is one of the most commonly used medications for the treatment of opioid addiction. However, as an opioid, it carries a potential for dependence and addiction which increases when this drug is abused or taken for a long time. Unfortunately, some people experience Subutex withdrawal symptoms after discontinuing its use.
If you’re trying to detox from Subutex, this can be devastating, and understandably so. Nonetheless, you shouldn’t let this demotivate you.
With professional help, you can overcome Subutex withdrawal symptoms and return to a life without opioid dependence quickly, safely, and effectively.
What Is Subutex?
Subutex is an FDA-approved medication commonly used to treat opioid use disorder (OUD) that contains buprenorphine, a semi-synthetic partial opioid agonist derived from thebaine. It comes in the form of a sublingual tablet and is typically administered once a day.
Subutex—also known by its street name —Subbies——should not be confused with Suboxone. While both are buprenorphine products, Suboxone also contains naloxone.
Subutex is primarily used in medication-assisted treatment (MAT). This treatment combines medications such as buprenorphine and methadone with behavioral therapy and counseling to help people recover from opioid addiction.
When taken as intended, buprenorphine reduces opioid cravings and the risk of opioid abuse and overdose. Relieving cravings and other withdrawal symptoms can help people come off stronger opioids.
As a partial opioid agonist, Subutex activates opioid receptors only to a certain degree and doesn’t produce the same level of euphoria as full opioid agonists like hydrocodone or oxycodone. It also has a “ceiling effect,” meaning that after a certain point, larger doses don’t amplify its effects.
While this minimizes the risk of Subutex abuse, it is still possible to misuse this drug (e.g., by crushing and snorting Subutex pills to enhance their effects) and become addicted.

Subutex Usage for Treating Opioid Dependence or Severe Pain
Subutex sublingual tablet contains buprenorphine, a partial opioid agonist, and is indicated for the treatment of opioid dependence and preferred for induction. Prescription use of this product is limited under the Drug Addiction Treatment Act.
Subutex is a synthetic opioid drug and a partial, long-acting µ-opioid receptor agonist. This means it activates the opioid receptors slightly less than a full agonist like heroin. Subutex also has effects on the κ- and δ-opioid receptors. Due to its partial agonist pharmacological profile, the impact of Subutex plateaus at high doses. This is associated with a more beneficial safety profile.
Subutex is one of the drugs widely used for medication-assisted treatment of opioid use disorders such as heroin addiction. During medication-assisted opioid treatment, the ingredient buprenorphine substitutes itself for the stronger opioid on which the patient is dependent.
Over time, the Subutex dose is lowered over a careful tapering schedule. In this capacity, buprenorphine has been included in the WHO list of essential medications. Buprenorphine is also a potent analgesic used for the short and long-term treatment of severe pain not adequately controlled by non-opioid drugs.
Subutex Withdrawal Symptoms
You may experience Subutex withdrawal symptoms, which can be both physical and psychological if you stop taking this medication too quickly.
Like any other opioid, buprenorphine can eventually lead to tolerance, dependence, and addiction. Opioids exert their analgesic and euphoric effects by binding to and activating the opioid receptors in the brain, which releases endorphins. Endorphins are the body’s natural method of pain relief. They can also promote feelings of well-being and euphoria.
With long-term Subutex use, the endorphin and opioid receptor system changes and adapts to a new functional level. Opioid receptors begin to respond to activation in a less pronounced manner, leading to the development of opioid tolerance.
In an attempt to get the same feeling as before, many patients begin to increase their Subutex dosage, leading to a dangerous spiral of opioid dependence and addiction. If you suddenly stop taking Subutex after becoming dependent on it, you’ll inevitably experience withdrawal symptoms.
Subutex withdrawal symptoms often look similar to those of other opioids and may include:
- Anxiety
- Bone, muscle, and joint pain
- Chills
- Depression
- Diarrhea
- Excessive yawning
- Fever
- Headaches
- Insomnia
- Mood changes
- Nausea and vomiting
- Opioid cravings
- Pupil dilation
- Sweating
- Watery eyes and runny nose
Buprenorphine can also cause precipitated withdrawal symptoms. These occur when Subutex is taken prematurely while another opioid is still in the system. This can be prevented by waiting for the onset of opioid withdrawal symptoms beforeadministering Subutex.
Subutex Withdrawal Timeline
How long Subutex withdrawal lasts depends on several individual factors—the Subutex dose taken, the duration of buprenorphine use, the patient’s health condition, etc. Whether or not you’re undergoing any Subutex detox treatment can also impact how long your symptoms last.
Let’s break down the typical Subutex withdrawal timeline by days:
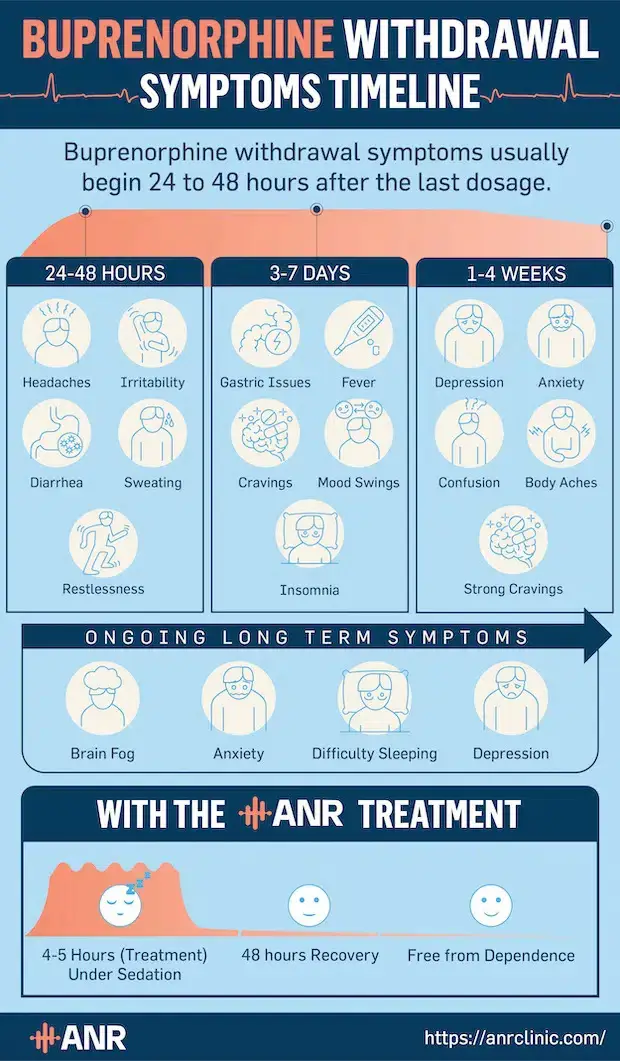
Withdrawals from Subutex tend to start relatively late—within 24–48 hours after the last dose—as it is a long-acting opioid. At this point, you may experience irritability, restlessness, diarrhea, and flu-like symptoms, such as runny nose and headache.
Physical Subutex withdrawal symptoms usually peak around the third day following buprenorphine discontinuation before slowly subsiding within 7–10 days. Fever, insomnia, mood swings, nausea, vomiting, and body aches are very common at this time.
As physical symptoms of Subutex withdrawal improve, the psychological ones—depression, intense cravings, and anxiety, among others—start to intensify. These symptoms usually last up to a month and may put you at a high risk of relapse and buprenorphine overdose.
Subutex Post-Acute Withdrawal Syndrome (PAWS)
Subutex post-acute withdrawal syndrome (PAWS) is an unpleasant phenomenon marked by protracted withdrawal symptoms.
For most people, buprenorphine withdrawal symptoms gradually fade away within more or less a month. For some, however, these symptoms may last several more weeks, months, or even years, exposing them to a high risk of relapse and affecting the overall quality of their lives.
Subutex PAWS is mainly associated with psychological withdrawal symptoms, such as:
- Intense opioid cravings
- Depression
- Anxiety
- Difficulty concentrating
- Apathy
- Mood swings
Although PAWS can cause you to lose hope in recovery, don’t let it discourage you. You can overcome any ongoing Subutex withdrawal symptoms you’re suffering from with medical help and return to a normal, healthy daily life.
How to Manage Subutex Withdrawal Symptoms
To manage Subutex withdrawal symptoms safely and effectively, seek help from medical professionals. Your doctor can explain how to get off Subutex correctly. To prevent uncomfortable withdrawal symptoms, they’ll likely suggest following a tapering schedule.
Under no circumstances should you quit Subutex abruptly, as this can trigger Subutex withdrawal symptoms. Tapering off opioids, including buprenorphine, allows your body to slowly adjust to lower doses of the drug.
Withdrawal from Subutex can be very unpleasant and painful. To minimize the risk of relapse and a potentially fatal opioid overdose, you should never detox from Subutex without medical supervision.

Be wary of Subutex detox centers that claim they can get you off buprenorphine in a couple of days. Rather than repairing the damage opioids cause to the endorphin-receptor system, Subutex detox simply helps remove the medication from your system.
Since it only treats the symptoms but not the cause of addiction, Subutex detox often leads to relapse. Due to lowered opioid tolerance, you may be at a greater risk of overdose, coma, brain damage, and death after detoxing from Subutex.
Risks and Dangers of Taking Subutex
As is the case with any opioid, taking Subutex puts you at risk of opioid abuse, dependence, addiction, and overdose.
Initially, Subutex (and its ingredient, buprenorphine) was marketed as a drug with a very low potential for abuse. With time, however, it became clear that it had its own potential for misuse. It is most frequently abused by injecting it intravenously or by crushing pills and then snorting Subutex powder.
A 2010 study found that slightly above 35% of patients receiving buprenorphine for opioid addiction treatment misused this medication by injecting it, with half of the patients doing this in an attempt to relieve opioid withdrawal symptoms. Less than 13% of these patients misused it to get “high.”
That aside, Subutex can cause various side effects, ranging from mild to potentially life-threatening. These include:
- Constipation
- Nausea
- Vomiting
- Dizziness
- Drowsiness
- Confusion
- Dry mouth
- Headache
- Respiratory depression
If you experience severe nausea and vomiting, whether as a Subutex withdrawal symptom or a side effect of the medication, seek medical help. If left untreated, this could lead to extreme dehydration and electrolyte loss, which can be fatal.
Subutex Rapid Detox
Subutex rapid detox is an anesthesia-assisted opioid detox method that can quickly flush buprenorphine out of your system, most often with the help of naloxone or another opioid antagonist. When performed under sedation, rapid detox diminishes the intensity of painful and uncomfortable withdrawal symptoms, but it doesn’t completely eliminate them.
The cost of Subutex rapid detox can go up to $24,000, but despite its hefty price, this treatment doesn’t ensure long-term recovery from opioids. In fact, it isn’t unheard of for patients to relapse soon after undergoing rapid Subutex detox, which can be life-threatening since the procedure decreases their opioid tolerance.
Not only is rapid detox from Subutex ineffective in treating opioid addiction, but it is also not safe. This procedure can lead to various complications and adverse events, including pulmonary edema, heart attack, and suicidal ideation.
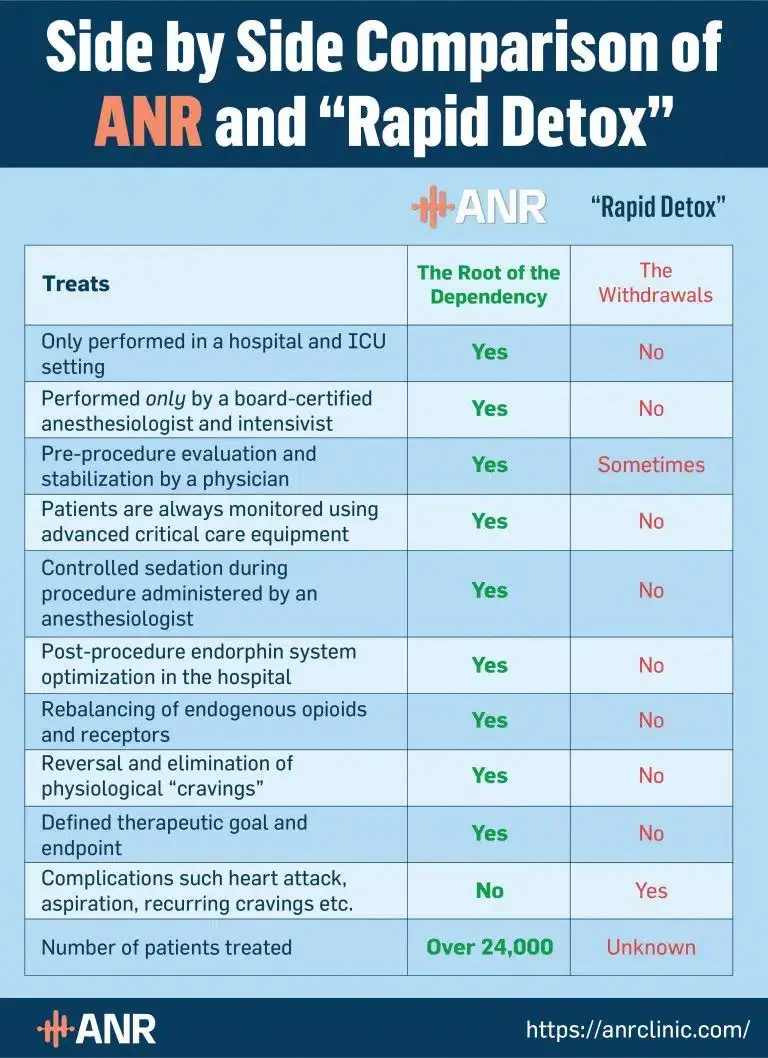
Subutex Rapid Detox vs. ANR Treatment
Accelerated Neuro-Regulation (ANR) is the only opioid addiction treatment designed to address the deep-rooted causes of opioid dependence. Unlike Subutex rapid detox, ANR negates the risk of ongoing withdrawal symptoms and relapse by re-regulating the endorphin-receptor system.
However, this isn’t the only difference between rapid Subutex detox and the ANR treatment. While the quality of rapid detox varies from one Subutex detox center to another, ANR is always performed in accordance with the highest safety and quality standards. Unlike rapid detox, it is carried out in an ICU setting of accredited hospitals by board-certified medical professionals.
Not to mention, each ANR treatment is personalized to suit every patient’s individual needs and medical history. Thanks to this, it is very safe—even for those with complex medical conditions—and doesn’t lead to complications.
Here’s a more detailed comparison of rapid Subutex detox and the ANR treatment:
ANR Opioid Treatment for Subutex Addiction
ANR is the safest and most effective opioid dependency treatment that can help you recover from Subutex addiction.
The ANR treatment goes beyond detox and withdrawal symptom management that most other opioid dependency treatments, including rapid detox, aim to achieve. For ANR patients, these are just the first steps toward an opioid-free life.
What follows afterward is the modulation of the opioid and endorphin-receptor systems, which eliminates cravings and the risk of relapse. Consequently, ANR is the only treatment option that addresses the root cause of opioid dependency by returning the brain to its normal state.
Not only that, but ANR doesn’t replace Subutex with other potentially addictive medications. Unlike other treatments, it enables you to resume your regular life and leave opioids in the past right after a short hospital stay.
To begin your recovery journey with ANR, schedule a free consultation with us today!
Key Takeaways
If you want to quit Subutex, consult with your doctor. Don’t quit this medication abruptly, as this increases the risk of Subutex withdrawal symptoms and relapse, among other negative consequences.
Here are the main things about Subutex withdrawal and detox you should remember:
- Even though Subutex is used to treat opioid addiction, it can lead to dependence and withdrawal symptoms—especially when abused.
- Subutex withdrawal symptoms include the typical symptoms of opioid withdrawal: fever, runny nose, nausea, diarrhea, cravings, depression, vomiting, etc.
- Without treatment, Subutex withdrawal can last a month and even longer. The ANR treatment can help you recover from Subutex addiction in a matter of days.
Subutex Withdrawal & Detox FAQ
1. How does Subutex affect the brain?
Subutex affects the brain by attaching to opioid receptors, which prevents other opioids from binding to them. When taken as intended, it relieves opioid withdrawal symptoms, including cravings, without inducing the typical euphoria associated with opioid use.
2. What are the signs of Subutex use disorder?
Tell-tale signs of Subutex use disorder include abusing the medication (e.g., snorting Subutex or taking it without a prescription) and experiencing intense cravings or other withdrawal symptoms after discontinuing its use. People with a Subutex use disorder will also typically spend a lot of time obtaining, using, and recovering from buprenorphine.
3. What is ANR opioid treatment?
ANR is an abbreviation for Accelerated Neuro-Regulation, which is a revolutionary opioid addiction treatment that reverses opioid-induced brain changes. By restoring balance in the endorphin-receptor system, ANR eliminates opioid addiction, together with the risk of relapse.
4. What is Subutex used for?
Subutex is used for the treatment of opioid use disorder (OUD), most often in medication-assisted treatment. It is one of the brand names for buprenorphine, a partial opioid agonist that can also be used to relieve moderate to severe pain.
5. Can Subutex cause opioid addiction?
Subutex can cause opioid addiction, as it is an opioid medication. While Subutex addiction is less common than that of other opioids, abusing Subutex and taking it for an extended time can increase the risk of becoming addicted to it.
6. How long does Subutex withdrawal last?
Subutex withdrawal typically lasts up to a month. Physical withdrawal symptoms usually subside within the first 10 days after quitting Subutex, but psychological symptoms may linger for several weeks. That said, how long Subutex withdrawal lasts depends on various individual factors ranging from the extent of your opioid use to your metabolism.
7. How long does it take to detox from Subutex?
How long it takes to detox from Subutex largely depends on the Subutex detox treatment you use, if any. Without any treatment, Subutex detox often lasts around a month. With the ANR treatment, you can bypass the withdrawal process altogether and make a long-lasting recovery from Subutex addiction within days.
